This blog is based on a webinar hosted by Vitaccess. For access to this webinar, and all webinars that we have hosted, please visit our dedicated webinars page.
What is a “micro-moment”?
During a clinician visit, you are much more likely to have accurate recall of a single, major change to your condition than you are of, for instance, day-to-day fluctuations in pain or sleep quality. These types of events, which are typically short, discrete, and variable in nature, can be referred to as “micro-moments”. Individually, micro-moments may be manageable – however, their cumulative impact on quality of life over time can be significant, and the challenge in their recall at clinician visits poses a major problem for their management.
Why is it valuable to capture micro-moments through real-world data?
Capturing and quantifying micro-moments through the collection of real-world data (RWD) can be valuable for a range of reasons: It helps to counteract the challenges of patients relying on their memory. Clinicians are not seeing these events, meaning that they cannot account for the full burden of disease when developing treatment programs. Capturing micro-moments solves the need to provide more granular data on quality-of-life impact.
Case studies
At Vitaccess, we have experience in developing a range of real-world studies to capture and quantify micro-moments effectively for the disease area of interest. Here, we provide two examples.
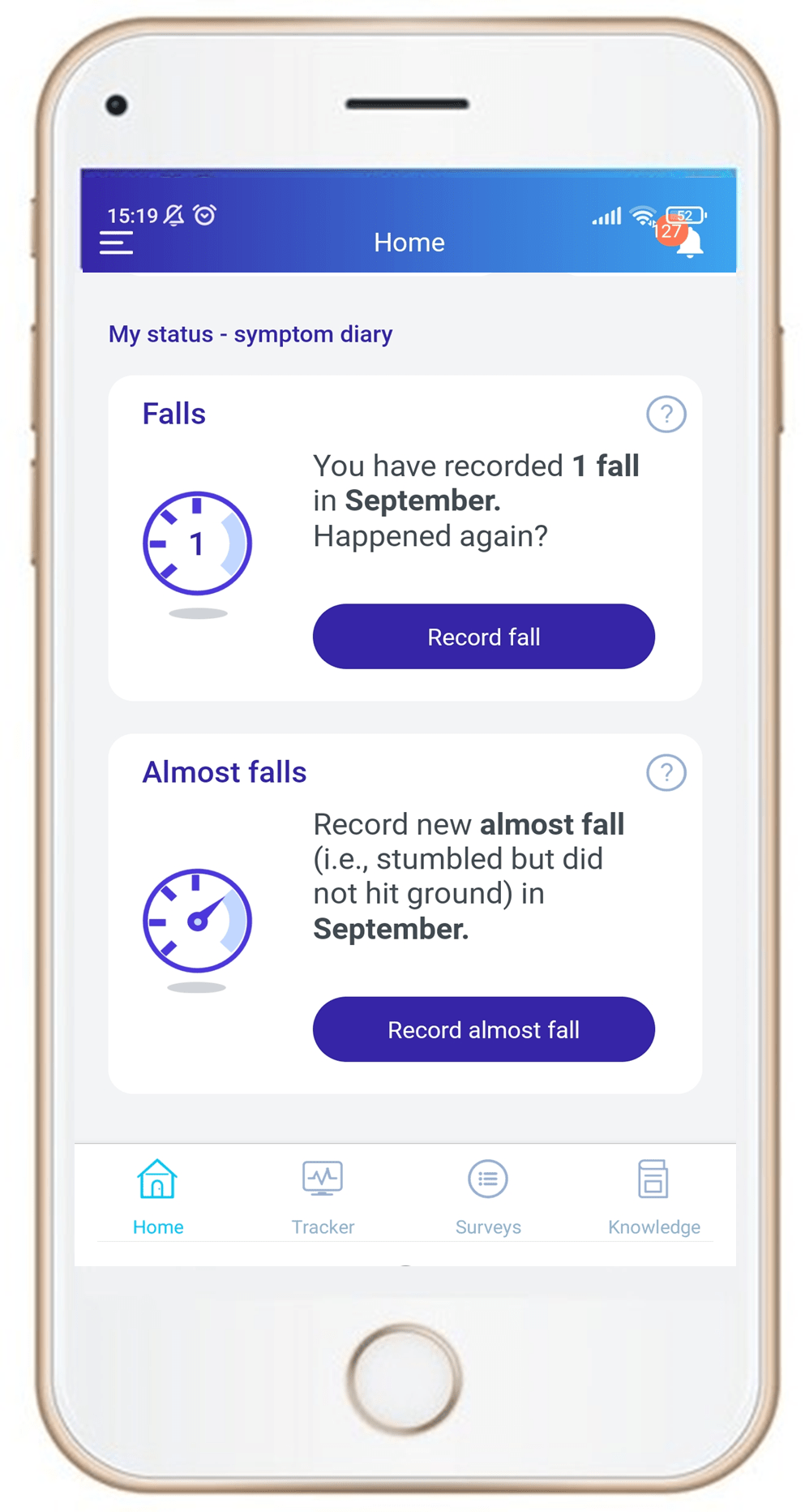
Charcot-Marie-Tooth disease (CMT)
- People living with CMT experience increasing problems with mobility and dexterity over their lifetimes, resulting in falls or near-falls. Our client for the ongoing CMT&Me study wanted to build a picture of this by capturing proximal micro-moments; to this end, we were able to use the Vitaccess Real™ platform to flexibly implement a counter in the study app, which participants can use to record falls or near-falls. Participants can then share the recordings of these events with their clinical teams to build a history of their experiences with falls.
Transfusion-dependent ß-thalassemia (TDT)
- TDT is a rare genetic disease resulting in anemia and the reliance on regular blood transfusions. To quantify the impact of transfusion as experienced by patients, we worked with our client to develop the bespoke myThalLog study app, which was used for daily data capture from participants over a 3-month period. We were able to implement a rewards program to encourage daily data input, which in turn allowed the client to build a comprehensive picture of transfusion burden over time.
Collecting RWD to adequately demonstrate disease burden, including micro-moments, can be vital to optimize drug development and therapeutic strategies. If you are interested in how we can help you to design and develop effective real-world digital studies, please contact us at info@vitaccess.com.
By Fatemeh Amini, Mark Larkin & Sally Vincent

 Copy Link
Copy Link


